Q & A With Psychological Scientist Linda Bartoshuk
 APS Past President Linda Bartoshuk is a leading taste researcher at the University of Florida. We invited our Facebook and Twitter followers to ask Bartoshuk questions about her research – here is what she had to say:
APS Past President Linda Bartoshuk is a leading taste researcher at the University of Florida. We invited our Facebook and Twitter followers to ask Bartoshuk questions about her research – here is what she had to say:
Is there a link between supertasters and people who have phantom taste, since the more taste buds you have the stronger some tastes are?
This is a wonderful question and I wish we had the data to answer it. Phantom tastes are created in the brain by release of inhibition. That is, normally taste input from one taste nerve inhibits input from other taste nerves. If taste from one nerve is damaged (or anesthetized), activity in the disinhibited area goes up leading to the sensations we call “phantoms.” I would guess that supertasters will have more intense phantoms, but we don’t have enough data to be sure. This should be addressed in future studies.
Would you rather be a “supertaster” with all of the risks, or have no taste at all?
I would much rather be a supertaster than have no taste at all because having no taste is extremely distressing to patients. Imagine never tasting sweet or salty again. The love of these tastes is hard wired in the brain to insure that we get critical nutrients (sweet signals sugars – brain fuel; salty signals sodium – key mineral in nerve and muscle function). Loss of taste thus means loss of a very important source of pleasure. By the way, supertasters (although they have to tolerate some nasty bitter tastes) experience more extremes of pleasure from food. One of our students in Food Science, Jaclyn Kalva, asked supertasters and others to rate the pleasure they get from their favorite food. Supertasters rated their pleasure significantly higher than did others.
It was mentioned that ear infections can harm taste, how so? And does it only affect the bitter tasting receptors or all of them?
The chorda tympani taste nerve carries taste information from the entire anterior tongue (the part you can stick out). That nerve passes through the middle ear on its way to the brain. Middle ear infections (called “otitis media”) can damage that nerve (Gedikli et al., 2001). Bitter fibers have the smallest diameters and are typically unmyelinated (myelin is a layer around nerve fibers). This means that pathogens (e.g., viruses, etc) can probably invade these taste nerve fibers more easily than they can invade other taste nerve fibers. This may explain why we have seen quite a bit of damage to bitter taste in people who have had a lot of ear infections. However, the taste damage from ear infections can affect other taste qualities as well (Bartoshuk, Duffy, Reed, & Williams, 1996).
Is there an easy at home test to check if we are super-tasters?
There are two ways to get a pretty accurate classification at home. First, think about a scale from zero (no sensation) to 100 (most intense sensation of any kind you have ever experienced) (Bartoshuk, Fast, & Snyder, 2005). Now think about the strongest taste sensation you have ever experienced. If the strongest taste intensity is greater than 70, you are likely a supertaster. There is also an anatomical way to tell (Bartoshuk, Duffy, & Miller, 1994). Swab blue food coloring on your tongue. Then look at your tongue in a magnifying mirror. You will see circles (maximum diameter about 1 mm but most are smaller) that are lighter blue than the rest of your tongue. These lighter circles are fungiform papillae, the structures that house taste buds. Create a template: a circle 6 mm in diameter. This is about the diameter of a hole punch like that used to make holes in notebook paper. Place the template on the tip of your tongue so that the circle touches the midline of your tongue and one edge. Now count how many fungiform papillae can be seen in the template. If your have more than 35, you are likely a supertaster. The most extreme supertaster I have seen had 60 fungiform papillae in the template.
It seems that it is easy to see that the tongue map was wrong, and had no data supporting it, how did it get so widely used?
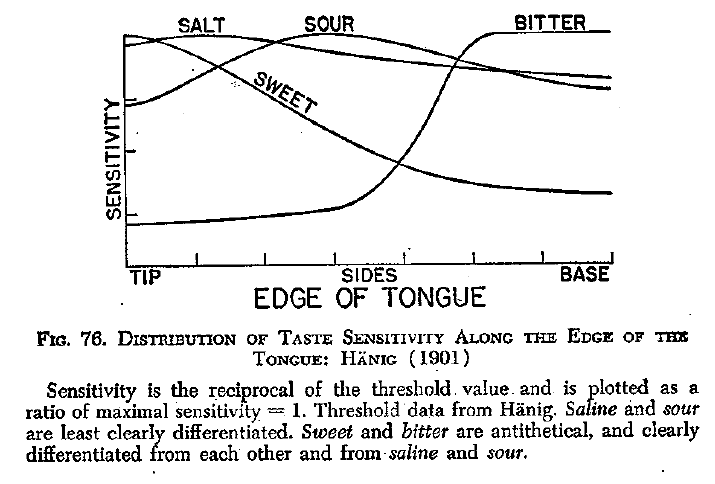
Edwin G. Boring: "Sensation and Perception in the History of Experimental Psychology"
The original error was made by Edwin Boring, professor of Psychology at Harvard who translated early psychology articles written in German (L. M. Bartoshuk, 1993; L.M. Bartoshuk, 1993). In his Sensation and Perception in the History of Psychology (Boring, 1942), Boring discussed the work of Hänig, who had been a student of Wilhelm Wundt in Leipzig. In 1901, Hänig had written a paper in Philosphishe Studien, “Zur Psychophysik des Geschmackssines” (Hänig, 1901). In that paper he measured taste thresholds around the perimeter of the tongue. He wanted to show variation in the thresholds that would lend support to the argument that the four basic tastes were mediated by different mechanisms. Although this seems silly today, it was an important question in 1901. Hänig found variation but it was very small. That is, if one translates the German correctly, the variation can be seen to be very small. Boring transformed the data; I suspect that he had trouble understanding the concentration units so he avoided them by calculating the reciprocals of the thresholds and calling this “sensitivity” and then reporting the values as ratios of maximal sensitivity. But as Boring’s Figure 76 shows, this lost all information about the magnitude of the original threshold values. The figure makes bitter look maximal at the base of the tongue and essentially zero at the tip. Sweet looks the opposite: near zero at the base and maximal at the tip. Salt and sour showed little variation around the perimeter of the tongue. Later writers just simplified the story with a picture of the tongue showing sweet at the tip, bitter at the back. Strangely enough, salt and sour are usually placed on the sides in such pictures. The ease of just including such a picture in taste chapters is probably the main reason this became dogma about taste.
Some years ago I gave a lecture to a grade school class that had done a taste lab testing the tongue map. I asked the students if they had gotten the answer in the book (the erroneous tongue map). They replied that they had not. I asked them why they thought they did not get the answer in the book. The answer: “We must have done it wrong.” The students were more willing to accept the authority their book represented than their own observations.
Is the tongue map still being put in today’s textbooks?
The map is still in some, but most texts now have much more up-to-date sections on taste. I have been trying to get tongue maps out of psychology texts for years. The most successful strategy was to visit publishers’ booths at psychology conventions and point out the error.
I’ve noticed that foods I used to hate, I now like – Do your taste buds change over time?
Yes, taste buds do change over time, but that is not the only reason you may like foods now that you used to hate. First, let me discuss taste bud changes over time. One of the best-documented changes is the loss of bitter with age (Prutkin et al., 2000). Actually, we do not know whether age is the variable or whether you just get more opportunities to damage bitter taste as time cumulates. Bitter taste is mediated by small unmyelinated fibers that are easily damaged. For example, viruses can invade these fibers and even the common cold can diminish the ability to taste bitter. There is also a sex difference with the loss of bitter. For males, bitter taste is lost across the adult age span. This may be the accumulation of pathologies. For females, there is a drop in the ability to taste bitter at menopause. We do not know the mechanism, but it makes biological sense if we understand that bitter is used by nature as a cue for poisons. The most critical time to want women to detect poison is during child-bearing. Second, our preferences change with time through conditioning. There are certain substances that we need and that our brains recognize in our diets. These include salt (sodium is essential for nerve and muscle function), sugar (glucose is brain fuel), fat (we need calories to survive) and protein (we need protein to provide the amino acids necessary to build our muscle tissue). When we eat foods that contain these substances, our brains detect these events. Salt and sugar stimulate receptors in the mouth that signal the brain; fats and proteins are broken into their constituents in the stomach and those constituents, fatty acids and glutamate (coming from one of the amino acids that make up proteins), signal the brain. When these signals arrive at the brain, the brain increases the palatability of the foods that gave rise to the signals. Over time, you like these foods more and more. Note that the key elements of junk foods (salt, sugar, fat) are substances that are crucial to survival when we are young. But with time as these foods become increasingly palatable overconsumption leads to chronic disease.
Do your taste buds adjust through repeated exposure? (ie spicy foods)
You do adjust to spicy foods like capsaicin (the substance that makes chili peppers burn), but the adjustment is not made by taste buds. Rather, the receptors that respond to hot spices are called “polymodal nociceptors” the oral burn produced by these receptors projects to the brain via the trigeminal nerve. These receptors can desensitize (Karrer & Bartoshuk, 1991). If you eat foods containing capsaicin frequently (like every other day), the burn will diminish through desensitization. However, this adjustment is only temporary. Take a vacation from eating chili pepper spices (cayenne pepper, hot sauces, etc) for a couple weeks. Then go back to your usual level of these spices. You will likely find them to be too hot. If you order a dish containing capsaicin in a restaurant and it proves to be hotter than you like, you can use desensitization to cool it off. Eat a good mouthful of the overly hot dish and then just sit and chat while the burn fades. Once the burn has disappeared (this may take 15 minutes or so) then continue to eat the dish. The burn level should be considerably reduced.
What causes the “metallic” taste experienced by some cancer patients? Why metal instead of any other taste? Is it connected at all with different chemotherapy chemicals filling the body?
Some compounds actually taste metallic (Stevens, Smith, & Lawless, 2006). There are several possible sources for the metallic tastes reported by some cancer patients. First, drugs can diffuse from blood into saliva. Drugs can also enter the mouth through crevicular fluid (the fluid that accumulates in the gums); chewing can express this fluid into the mouth. Drugs can also stimulate taste through a mechanism called “venous taste” (Matsuyama & Tomita, 1986). Taste buds are clusters of taste receptor cells. Only the tips of the receptor cells are in contact with the mouth. The rest of the taste bud is sealed off. However, there are receptor sites on the bottoms of the receptor cells in the taste bud. Drugs can diffuse out of blood and stimulate those receptor cells without ever entering the mouth. Some patients can taste those drugs. We have few data on this, but the small number of patients I have seen who taste their chemotherapeutic drugs have been supertasters. This is a great area for future study.
There is another mechanism for metallic taste that is related to the taste damage that chemotherapy can cause. Taste is mediated by three cranial nerves (CNs VII, IX, X). These nerves inhibit each other in the brain such that damage to one of them releases inhibition on the others (Halpern & Nelson, 1965; Lehman, Bartoshuk, Catalanotto, Kveton, & Lowlicht, 1995). That release of inhibition intensifies signals in the brain areas receiving input from the undamaged nerves. This intensification compensates for the loss of input from the damaged nerve. This constancy mechanism preserves whole mouth taste experience. This system appears to operate more generally as well. The inhibition generated by taste input also serves to diminish activities that might interfere with eating. Failure to eat will lead to starvation and death so nature protects eating behavior. For example, if an animal were to experience a tongue injury, the resulting pain might prevent eating and cause the animal to starve. However, the inhibition produce by taste input could turn off oral pain while the animal ate. Unfortunately such mechanisms come with a cost. When taste damage releases inhibition on brain areas receiving input from undamaged cranial nerves, that release produces sensations. We call these sensations produced in the absence of normal stimulation “phantoms” (Bartoshuk et al., 2005; Yanagisawa, Bartoshuk, Catalanotto, Karrer, & Kveton, 1998). Metallic sensations are the most common taste phantoms experienced from the damage induced by chemotherapy although these phantoms can also be bitter, sour, salty or sweet. Incidentally, taste damage in supertasters can result in severe oral pain phantoms. This disorder is called “burning mouth syndrome” (Grushka & Bartoshuk, 2000).
Is it known what causes the taste nerve damage that causes phantom tastes?
Unfortunately, there are many sources of taste nerve damage that can cause phantoms:
• viral damage (ear infections, upper respiratory infections)
• damage from dental anesthetics (anesthetics like lidocaine are injected into a space between muscle and bone through which the nerve mediating pain travels; one of the taste nerves travels through the same space and can be damaged by the needle or the anesthetic),
• damage from extracting third molar teeth
• head injuries damage taste nerves where they pass through bone
• Surgery for acoustic neuromas (tumors of the auditory or 8th cranial nerve) can damage the near-by 7th and 9th cranial nerves (both mediating taste).
What can/do researchers do in order to make the scale more uniform so that two people’s perceptions of bitter for example, would be known to be equal?
The solution is to use a technique called “magnitude matching” (Bartoshuk, Fast, et al., 2005; Marks et al., 1988). We are very good at matching perceived intensities from different sensory modalities. For example, we can select a light that is as bright as a tone is loud (“cross-modality matching”). Similarly, we can select a tone that is as loud as a quinine concentration is bitter. We can use this methodology to study supertasters (individuals with the largest number of fungiform papillae, structures that house taste buds). We asked subjects to match the bitterness of PROP (6-n-propylthiouracil) to the loudness of a tone. Some subjects matched the PROP bitterness to a much louder tone than did others: these subjects turned out to be those with the largest number of fungiform papillae. Thus the key is to identify a standard: a modality unrelated to the modality of the sensation you want to compare across subjects. In our PROP experiment, we selected loudness of a tone because we believed the perception of loudness was unrelated to the perception of taste. We knew that perceived loudness was not equal to all but since it was not related to taste, the loudness of our standard would be, on average, equal to those with the most fungiform papillae and those with the fewest.
I think that some current problems with sensory scaling also reveal how hard it is to argue against a generally accepted position. I read a paper by a graduate student who had used a Likert scale to claim that two different groups experienced different sensations (Likert scales cannot be used to make sensory comparisons across individuals or groups) (Bartoshuk et al., 2002). I pointed out her error and she agreed with me. But she went on to tell me that the journals would accept her Likert scale data because everybody used the scale and she needed to publish the paper. If a mistake is made often enough it takes on authority that is hard to overcome.
If MSG isn’t really a taste enhancer, why do so many food companies add it to foods?
MSG (monosodium glutamate) is essentially a condiment; that is, it is used to make foods more palatable because many people like its taste. Thus the key question is why many like it? We used to think that taste receptors were only in the mouth. The “basic tastes” sweet, salty, sour, bitter had biological utility; their hard-wired affect solved critical nutrition problems quickly before an organism had a chance to learn about its environment. Thus a newborn could nurse immediately because it was born liking the sweet taste of mother’s milk. A newborn could also avoid most poisons because it was born disliking the bitter taste characteristic of many poisons. Olfaction solved nutritional problems that we had time to learn. Thus a rat pup could learn to like to eat a particular grain by sniffing it on its mother’s breath (Galef, 1979). However, recent research has revealed greater complexity. Taste receptors are not only in the mouth but also throughout the digestive tract. Our mouths can tell us to consume the sugars in mother’s milk and the sodium salts necessary for nerve and muscle function. Our mouths can also tell us to avoid bitter poisons. But nutritionally we need fats and proteins; how does nature make certain we get them? As it happens, fat and protein molecules are very large complex molecules – too large for either taste or smell so our mouths cannot tell us to eat them without some help. Nature took advantage of the receptors in the digestive tract to make sure we will learn to like to eat sources of fat and protein. Both fats and proteins are broken into their constituent parts by digestion. There are receptors for those constituent parts (fatty acids from fats, the amino acid glutamate from proteins) in the digestive tract. Thus the stomach can signal the brain that you have eaten a food containing fat or protein. Our brains make us like the sensations evoked by the foods that gave rise to the fatty acids and glutamate; we call these “conditioned food preferences.” Note that food companies can add glutamate to any food and it will lead to a preference because the glutamate will stimulate the glutamate receptors in the digestive tract. The signal in the mouth (there are glutamate receptors in the mouth) will make the taste of glutamate itself liked. Unfortunately for fatty acids, although there are fatty acid receptors in the mouth, fatty acids taste terrible and do not themselves become liked.
Is there a difference between genders when it comes to taste?
Yes, women are more likely than are men to be supertasters. Thus, on average, women experience more intense taste sensations than do men. Incidentally, supertasters (and thus women) have more intense liking for their favorite foods and more intense disliking for their least favorite foods. In addition, as we noted above, there are differences between males and females with age.
Does the sensory input coming from your taste buds change over time or is it the perceptual output that changes over time?
Both. As noted in an earlier question, there are genuine changes in our taste buds. However, let me enlarge on this answer. When we talk about “taste” in our everyday lives, we really refer to the sensations evoked when we eat. This includes olfaction, particularly retronasal olfaction. The nose actually has two biological functions. When we sniff odors from our environments, we pull them into our nostrils and they eventually make their way to the olfactory receptors at the top of the nasal cavity (orthonasal olfaction). When we chew and swallow foods, odors emitted from the foods are forced up behind the palate and into the nose from the rear. The odors then make their way to the same olfactory receptors (retronasal olfaction). Orthonasal olfaction tells us about odors in the world; retronasal olfaction tells us about the odors emitted by the foods we are eating. The combination of true taste and retronasal olfaction is called “flavor.” Here’s where strange word usage comes in. We can use “taste” as a verb; we taste food. We cannot use “flavor” as a verb. If we say we flavor food, this describes the addition of flavor to food not the perception of flavor. However this came about, it makes us use the term “taste” both to refer to pure taste itself and also to refer to the perception of flavor. Thus we can look at changes in “perceptual output” in the context of the broader meaning of “taste.” Our perception of taste changes over time in complex ways because taste and olfaction have such different contributions to make to the experience of eating.
References
Bartoshuk, L. M. (1993). Genetic and pathological taste variation: what can we learn from animal models and human disease? Ciba Found Symp, 179, 251-262; discussion 262-257.
Bartoshuk, L.M. (1993). The biological basis of food perception and acceptance. Food Quality and Preference, 4, 21-32.
Bartoshuk, L.M., Duffy, V.B., Fast, K., Green, B.G., Prutkin, J.M., & Snyder, D.J. (2002). Labeled scales (e.g., category, Likert, VAS) and invalid across-group comparisons. What we have learned from genetic variation in taste. Food Quality and Preference, 14, 125-138.
Bartoshuk, L.M., Duffy, V.B., & Miller, I.J. (1994). PTC/PROP tasting: Anatomy, psychophysics, and sex effects. Physiology and Behavior, 56, 1165-1171.
Bartoshuk, L.M., Duffy, V.B., Reed, D., & Williams, A. (1996). Supertasting, earaches, and head injury: Genetics and pathology alter our taste worlds. Neuroscience and Biobehavioral Reviews, 20, 79-87.
Bartoshuk, L.M., Fast, K., & Snyder, D. (2005). Differences in our sensory worlds: Invalid comparisons with labeled scales. Current Directions in Psychological Science, 14, 122-125.
Bartoshuk, L.M., Snyder, D. J., Grushka, M., Berger, A.M., Duffy, V, B,, & Kveton, J.F. (2005). Taste damage: previously unsuspected consequences. Chemical Senses, 30 (suppl 1), i218-i219.
Boring, E.G. (1942). Sensation and perception in the history of experimental psychology. New York: Appleton.
Galef, B.G. (1979). Social transmission of learned diet preferences in wild rats. In J.H.A. Kroeze (Ed.), Preference behaviour and chemoreception (pp. 219-231). London: IRL.
Gedikli, O., Dogru, H., Aydin, G., Tüz, M., Uygur, K., & Sari, A. (2001). Histopathological changes of chorda typani in chronic otitis media. The Laryngoscope, 111, 724-727.
Grushka, M., & Bartoshuk, L.M. (2000). Burning mouth syndrome and oral dysesthesias. The Canadian Journal of Diagnosis, 17, 99-109.
Halpern, B.P., & Nelson, L.M. (1965). Bulbar gustatory responses to anterior and to posterior tongue stimulation in the rat. American Journal of Physiology, 209, 105-110.
Hänig, D.P. (1901). Zur Psychophysik des Geschmackssinnes. Philosophiche Studien, 17, 576-623.
Karrer, T., & Bartoshuk, L. (1991). Capsaicin desensitization and recovery on the human tongue. Physiology and Behavior, 49, 757-764.
Lehman, C.D, Bartoshuk, L.M., Catalanotto, F.C., Kveton, J.F., & Lowlicht, R.A. (1995). Effect of anesthesia of the chorda tympani nerve on taste perception in humans. Physiology and Behavior, 57, 943-951.
Marks, L.E., Stevens, J.C., Bartoshuk, L.M., Gent, J.G., Rifkin, B., & Stone, V.K. (1988). Magnitude matching: The measurement of taste and smell. Chemical Senses, 13, 63-87.
Matsuyama, H., & Tomita, H. (1986). Clinical applications and mechanism of intravenous taste tests. Auris Nasus Larynx, 13, S 43-S 50.
Prutkin, J.M., Duffy, V.B., Etter, L., Fast, K., Gardner, E., Lucchina, L.A., . . . Bartoshuk, L.M. (2000). Genetic variation and inferences about perceived taste intensity in mice and men. Physiology and Behavior, 69, 161-173.
Stevens, D.A., Smith, R.F., & Lawless, H.T. (2006). Multidimensional scaling of ferrous sulfate and basic tastes. Physiology & Behavior, 87(2), 272-279.
Yanagisawa, K., Bartoshuk, L.M., Catalanotto, F.A., Karrer, T.A., & Kveton, J.F. (1998). Anesthesia of the chorda tympani nerve and taste phantoms. Physiology and Behavior, 63, 329-335.




APS regularly opens certain online articles for discussion on our website. Effective February 2021, you must be a logged-in APS member to post comments. By posting a comment, you agree to our Community Guidelines and the display of your profile information, including your name and affiliation. Any opinions, findings, conclusions, or recommendations present in article comments are those of the writers and do not necessarily reflect the views of APS or the article’s author. For more information, please see our Community Guidelines.
Please login with your APS account to comment.